Free Water Knockout (FWKO): What You Need To Know
Free water knockouts (FWKO) are an essential component in the oil and gas industry, designed to remove any free water present in the production...

As simple as the media portrays the oil and gas industry sometimes, in reality, it is quite a sophisticated and elaborate process. With the many stages involved at a macro level, including drilling, production, and refining, the oil and gas process can be broken down to many more micro levels as well.
Keep reading to discover everything you need to know about the upstream oil and gas separation process.

Oil and gas separation is a common technique in upstream oil and gas wells, where crude oil and natural gas are often found in the same well. Oil and natural gas have different density levels, so when they mix together it can cause problems down the road. Oil and gas separation allows for these two products to be distinct from one another.
Oil is less dense than water, so it can float to the top. Generally, natural gas is much less dense than crude oil. Oil has a specific gravity of 0.85 g/cm 3, while natural gas has a specific density of about 0.55 g/cm 3. Gravity separation uses these differences in material to separate out different products found in one single stream into different streams that are distinct from each other, using mainly gravitational forces.
Process equipment companies in the oil and natural gas industry use a number of equipment units designed to separate, or "separate," crude oil from fluids involved in the production of oil. Oil and gas separation is done by physical means, not chemical means. The separation units are designed to accommodate an operating pressure range of 5-700 pounds per square inch (psi) that the fluids are under at the time of separation.
A separator is a vessel in which a mixture of fluids that are not soluble in each other are segregated from one another.
In the oilfield, separators are used to segregate gas from liquid; or one liquid, such as crude oil, from another liquid, such as water.
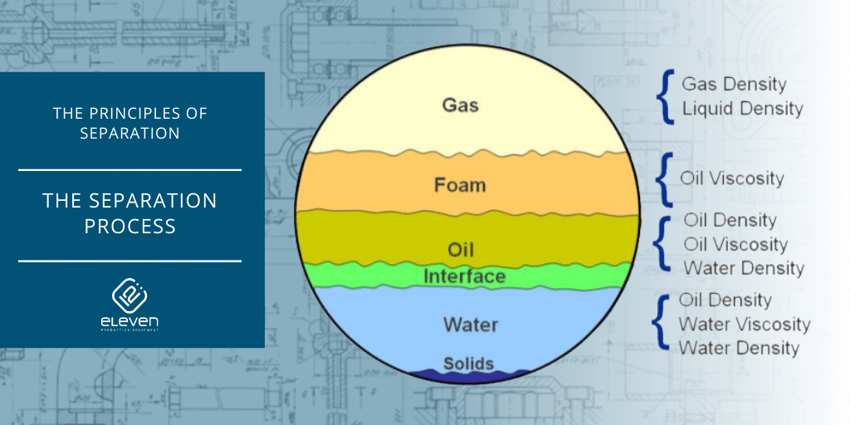
There are more separators on oil and gas locations or process facilities than any other type of process equipment. Sometimes they are called scrubbers, slug catchers, FWKO’s, centrifuges, and many other names.
Regardless of what the vessel is called, the function is to segregate two or more fluids (like when using shaker screens), usually gas and liquids.
There are seven principles that affect separation. In order to design a process for separating any mixture of oil and gas, these principles must be taken into account.
Depending on the properties of the well fluid and the required task at hand, these are the principles governing separation.
In some cases, only one of the seven may be used at any given time.
In other cases, it may be more expedient to use a combination of two or more of the principles to get the required results within a given timeframe.
This is the most important principle of oil separation. The pressure change required to separate a light oil from a heavy oil may be different, but there will always be some pressure change involved in separating two or more components of any mixture.
Depending on what needs to be done, pressure on a hydrocarbon containing vessel may be manipulated to enhance the separation process.
If the vapor pressure in a vessel containing hydrocarbons is decreased, some of the lighter hydrocarbons will flash from the liquid phase into the vapor phase.
 On the other hand, if vapor pressure in a vessel containing hydrocarbons is increased, some of the lighter hydrocarbons will condense from the vapor phase back into the liquid phase.
On the other hand, if vapor pressure in a vessel containing hydrocarbons is increased, some of the lighter hydrocarbons will condense from the vapor phase back into the liquid phase.
These statements are true if the temperature remains constant.
Temperature differences between fluids will affect their relative volatilities due to changing solubility and vapor pressure levels, which changes their equilibrium compositions during separation. Heavier oils have higher viscosity and lower vapor pressure than lighter oils at a given temperature, so they tend to accumulate near the bottom of an oil/water emulsion when the temperature is increased, resulting in improved separation efficiency compared to operating at low temperatures where both phases are more volatile.
For example, if a separation occurs by heating oil from refinery streams using heat exchangers before sending it into a thermal separator tower equipped with steam coils through which cooling water flows, the vapor being produced from the lighter oil will have a lower boiling point than that of heavier oils at a given temperature.
Temperature changes go hand-in-hand with pressure changes when it comes to separation in a hydrocarbon containing system or vessel.

If the pressure in a vessel remains the same, a rise in temperature will cause some of the lighter hydrocarbons to flash from liquid to vapor phase.
Likewise, a drop in temperature at a fixed pressure will cause some of the hydrocarbons to condense into the liquid phase.
Gravity, or more specifically, the difference in specific gravity of the components being separated is the biggest factor in the time it takes for the components to separate.

The greater difference in the specific gravity of the components, the faster separation will occur.
There is a significant difference in the specific gravity between gas, oil and water, so the gas does not take long to break out and rise to the top of the liquids.
The less difference between the specific gravity of the components means there must be more settling or retention time for the components to separate out. If there is a greater difference in the specific gravity of the components, such as in the gas and oil example, gross separation will take place rather quickly.
Scrubbing action is what separates oil from gas. The number of physical stages required to remove the oil is determined by the stripping action. Viscosity, pressure, temperature, and density are properties that can be used to determine which process will yield the most efficient separation.
Chemical action between two immiscible liquids is necessary for separation. The interfacial area between the two liquids must be retained to provide the chemical action. A decrease in temperature will always reduce or eliminate three-phase contact and therefore reduces separation efficiency.
Pressure has an inverse effect on separating efficiency. Decreasing pressure increases the relative velocity of separation, thereby increasing efficiency. Increasing pressure decreases relative velocities, reducing separating efficiency. Varying the speed of agitation changes the width of a contacting zone, which can affect separation efficiency either positively or negatively depending upon conditions.
Electrical separation is based on the fact that oil and water have different dielectric constants. When a mixture of oil and water is electrically grounded, so one phase has a positive charge and the other phase has a negative charge, the two phases will separate.
This method can be employed in both vertical and horizontal vessels.
Retention time of the oil in the separator can be used to determine how much gas can be recovered. The greater the retention time, the more oil that will be removed. This is due to the fact that as long as oil remains in contact with inert gas, it will not rise. The longer the contact time between the inert gas and liquid, the larger amount of oil will dissolve into the vapor phase.
Click below to Download our eBook containing the remaining five principles of the oil and gas separation process.
Also, learn more about the key factors to consider for separator optimization.
Discover the strengths that the 12:eleven specialists in innovation and customization bring to production and process equipment (including oil ang gas production separators). Contact 12:eleven and experience quality that is reliable, efficiency that is proven and service that is unsurpassed.
We invite you to request a quote for our products or services.
Oil and Gas Production and Process Equipment - In this blog post we will dive into the most important pieces of oil and gas production equipment. We will also talk about how to maintain them properly.
Oil Stabilizing Towers: What You Need to Know - In this article, we'll discuss the basics of oil stabilization towers and provide information on how they work, and the benefits they offer.
What You Need to Know About Gas Dehydration Units - Here's everything you need to know about gas dehydration units and how they can be used to eliminate unwanted moisture from recovered natural gas.
Everything you need to know about Glycol Contact Towers - In this post, we'll provide an overview of what a glycol contact tower is, explain the benefits of using one, and the types available.
As a specialty company providing custom design, engineering and fabrication of production and process equipment, we are dedicated to doing what’s right for our customers and passionate about creating value on every project.
We leverage the field expertise of our design and engineering group together with our diversified manufacturing capabilities to deliver a broad range of innovative surface production equipment - including Separators, Treaters, Free Water Knock Outs, Bulk and Test Packages, Indirect Line Heaters, Gas Production Units, Gas Dehydration Units, Glycol Contact Towers, Vapor Recovery Towers, Oil Stabilizing Towers.

Free water knockouts (FWKO) are an essential component in the oil and gas industry, designed to remove any free water present in the production...

Gas Production Units (GPUs) are a crucial component of the oil and gas industry. They play a significant role in the extraction, processing, and...

Piping modules and spools are an important part of any piping system. They help to keep the fluid in the system moving by providing a path for it to...